In this video, we're going to take a look at hydrohalogenation reactions. So under this type of reaction, one hydrogen and one halogen, either bromine or chlorine, are added to one pi bond. Here in our general type of reaction, we have an alkene to start with, and we're reacting it with HX. Here, one hydrogen would go to one of these double-bonded carbons and the halogen to the other. Both of them are the same; it's a symmetrical molecule, both alkene carbons have the same number of hydrogens. So here you could either add the H here or here, and then you'd add the halogen to the other side. Here I choose to add them in these positions, and at the end of it, what do we just make? Well, we transitioned from an alkene reactant to an alkyl halide product. So when it comes to hydrohalogenation, we're trying to create alkyl halide products as our final answer.
Hydrohalogenation Reaction - Online Tutor, Practice Problems & Exam Prep
Hydrohalogenation Reaction Concept 1
Video transcript
Hydrohalogenation Reaction Example 1
Video transcript
Complete the following hydrohalogenation reaction. So here we have cyclopentene reacting with hydrobromic acid. Here we're going to say, we're going to break the double bond, and both of these double bonded carbons are symmetrical, so the H could go to either one, and the Br could go to the other. Here I decide to add the hydrogen here, and then the bromine will go here. In the process, we've just created an alkyl halide. Here we've created bromocyclopentane as our final product.
Markovnikov's Rule Concept 2
Video transcript
Now the addition of hydrogen and a halogen to a non-symmetrical alkene or alkyne follows Markovnikov's rule. Under Markovnikov's rule, the hydrogen atom is added to the carbon, whether it be double bonded or triple bonded, with more hydrogens. And then the addition, and then the halogen atom or X atom is added to the triple bonded or double bonded carbon with fewer hydrogens. If we take a look here, we have an alkene to start, the alkene on the left only has 1 hydrogen, and the one on the right has 2. Following Markovnikov's rule, hydrogen would go to the double bonded carbon with more hydrogens, so it'd go here, and then the halogen would go to the one with fewer hydrogens, so it'd go here. In this process, we made an Alkyl Halide. Now, when we have an Alkyne, we have 2 π bonds, so we'd need 2 moles of HX. Still following Markovnikov's Rule, hydrogen would go to the now triple bonded carbon with more hydrogens, and there'd be 2 of them adding, and then we'd have 2 halogens adding to the triple bonded carbon with fewer hydrogens. So this one here. As a result of using 2 moles of HX, we wind up with a dihalide with both halogens adding to the same formally triple bonded carbon. So just remember, we use Markovnikov's rule if our double or triple bonded carbons have a different number of hydrogens each. Use this rule to determine what your final answer will be.
Markovnikov's Rule Example 2
Video transcript
Complete the following hydrohalogenation reaction. Here we have our alkene reacting with HCl. What we do first is we look to see what kind of double bonded carbons we have. The one on the left is making 4 bonds, so it has no hydrogens. The one on the right is making 3 bonds that we see, so it has 1 hydrogen. Since the two double-bonded carbons have different numbers of hydrogens, we are going to use Markovnikov's rule to determine where the H and the Cl will go. Following Markovnikov's rule, it states that the hydrogen will go to the double-bonded carbon with more hydrogens, so it would go here. Of course, the hydrogen that was originally there is still there, and then the chlorine would go to the double bonded carbon with fewer hydrogens. So it would go here.
This will represent the alkyl halide product formed from the hydrohalogenation between our reactants. Now remember, your hydrogens when they are connected to carbon are invisible, so you could erase them, and this would be our full skeletal formula for our product.
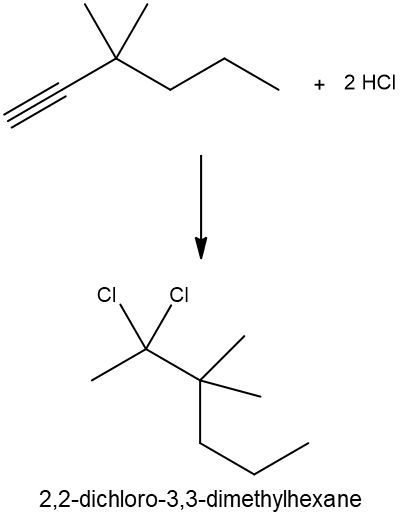
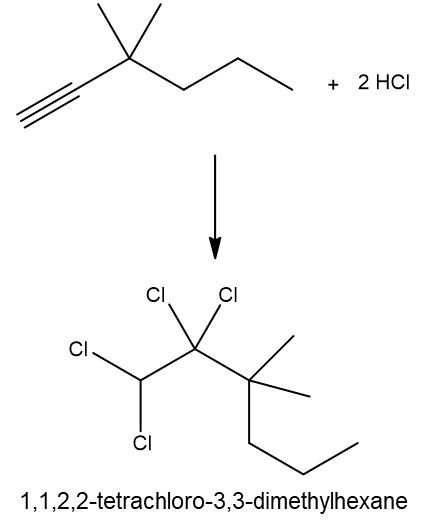
Write a hydrohalogenation reaction with excess HCl and name the organic product formed.
Do you want more practice?
Your GOB Chemistry tutor
- Ocimene, a compound isolated from the herb basil, has three double bonds and the IUPAC name 3,7-dimethyl-1, 3-...
- Draw all possible products formed when 2-methyl-2-butene undergoes addition with HCl. Label them as being eith...
- In the following addition reactions, are the given alkyl halides obtained as the major products? Give a reason...
- What alkene could you use to make the following products? Draw the structure of the alkene, and tell what othe...
- 1-Pentyne reacts with HBr in a 1:1 molar ratio to yield two different addition products, both being bromopente...
- Complete the following equations (Hint: Answers may include concepts learned from previous organic chapters):c...