In this reaction, we're going to talk about the halogenation reactions. Here, under this type of reaction, 2 halogens coming from either Br2 or Cl2 are added to 1 pi bond. So here we have our starting alkene, and we're reacting it with X2. In the process, each double-bonded carbon gains 1 halogen. At the end of this, we have a structure that has 2 halogens on it, which we call a dihalide. Now, if we have an alkyne that means we have 2 pi bonds. So remember, we need 1 mole of reagent for every pi bond. And since there's 2 pi bonds here, we'd need 2 moles of X2. The first mole would add, to give us 2 halogens, and then the second mole would add again, to give us another 2 halogens. At the end, you have a structure that possesses 4 halogens on it. So we'd call this a tetrahalide. So remember, halogenation is just adding 2 halogens per pi bond within your structure. This could give us a dihalide if you're dealing with an alkene, or a tetrahalide if you're dealing with an alkyne.
Halogenation Reaction - Online Tutor, Practice Problems & Exam Prep
Halogenation Reactions Concept 1
Video transcript
Halogenation Reactions Example 1
Video transcript
Complete the following halogenation reaction. So here we have our alkene, and we're reacting it with 1 mole of chlorine. Remember, all that's going to happen here is we're going to sacrifice our pi bond in order to add 2 halogens to my structure. Each double bonded carbon gets a chlorine. Here it doesn't matter the orientation that you place the chlorine if it's up or down or whatever, all that matters is the connection. It's these 2 double bonded carbons that each need to have their chlorines. Here I decide to show them as being on opposite sides of each other, but I could easily show them being on the same side. We don't go into spatial orientation when it comes to a reaction like this. That's left for when we do organic 1 and organic 2. For right now, this would be our dihalide structure completed from the following halogenation reaction.
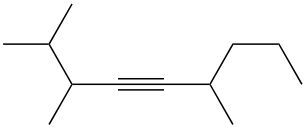
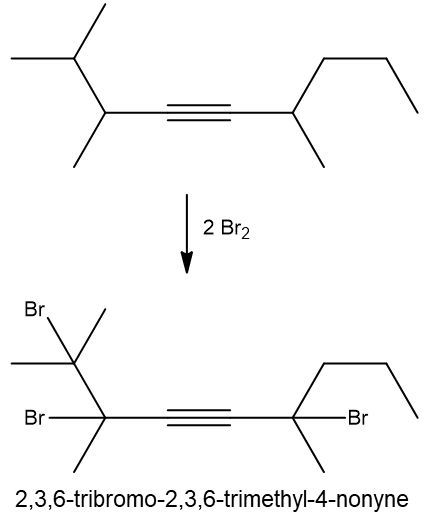
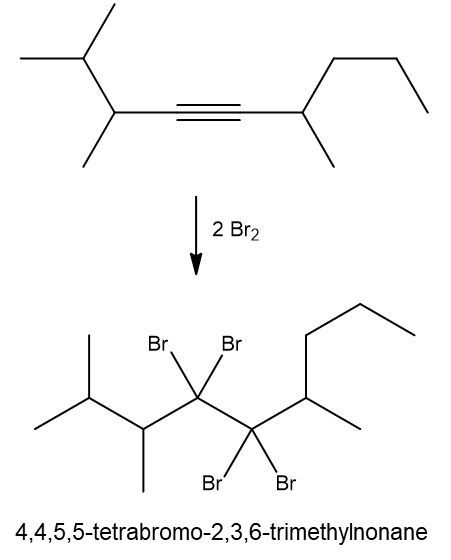
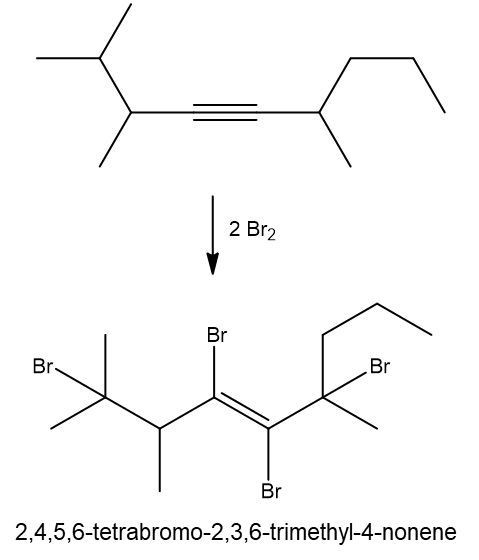
Write a halogenation reaction of the following alkyne with Br2 and name the product formed.
Do you want more practice?
Your GOB Chemistry tutor
- If bromocyclohexane were converted into cyclohexene, what kind of reaction would that be?
- 2,2,3,3-Tetrabromopentane can be prepared by an addition reaction of excess Br2 with an alkyne. Draw the struc...
- Identify the type of reaction for the following:a. <IMAGE>b. <IMAGE>
- What alkene could you use to make the following products? Draw the structure of the alkene, and tell what othe...