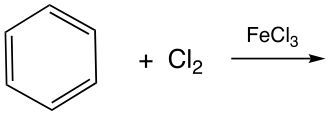Now we've seen benzene a few times within this chapter. Now benzene itself is what we call an aromatic compound. And aromatic compounds have an unusually or uniquely stable condition. Benzene possesses Pi bonds, but because of its aromaticity and its unique stability, it doesn't undergo additional reactions like we would expect. Benzene undergoes substitution reactions instead, and that's unlike alkenes and alkynes which do addition, because they have pi bonds, but they're not uniquely stable. Now here we're going to cover 2 types of substitution reactions that Benzene undergoes. One being Halogenation reactions, where we're substituting in a Halogen onto the Benzene ring, and then Friedel-Craft Alkylation, where we're substituting an alkyl group onto the benzene ring. So just remember, Benzene is pretty special. It's aromatic and very stable. It's gonna do substitution reactions instead of addition reactions, because it wants to maintain this very highly stable form that it possesses.
Benzene Reactions - Online Tutor, Practice Problems & Exam Prep
Benzene Reactions Concept 1
Video transcript
Halogenation Reactions Concept 2
Video transcript
In this video, we're going to take a look at the halogenation of benzene. Here, benzene would react with either Br2 or Cl2. In this reaction, one of the hydrogen atoms on the benzene is substituted with an X, which stands in for either bromine or chlorine. Here we'd say that a catalyst in the form of FeX3, where X3 is either Br3 or Cl3, must be used and has to contain the same halogen as the reagent. So basically, what I mean by this is if X2 here is Cl2, then this has to be FeCl3; if X2 here is Br2, then this has to be FeBr3. So, if we take a look here at this benzene halogenation reaction example, we have our benzene with its six hydrogens on it. Remember, X here equals either bromine or chlorine. What happens is that one of these hydrogens gets replaced by this halogen, one of them. And so here, the H is now gone, and instead in its place we put one of the halogens. That halogen could be chlorine or bromine. This is a way that we can substitute in a halogen at the expense of losing a hydrogen on benzene.
Halogenation Reactions Example 1
Video transcript
Of the following, which is a correct product of benzene substitution reaction with Br2? So remember, here we have the bromination of benzene, where we're going to substitute out a hydrogen on benzene for a bromine. Option A doesn't work because we substituted in an iron, which doesn't work. Option B doesn't work because we only substitute in 1 bromine, not 2. Option C doesn't work because the hydrogen is still there. The answer here is option D because there was a hydrogen here, and we substituted it out to get the bromine there, creating bromobenzene as our answer. So here option D is the correct answer.
Name the product of the following benzene halogenation reaction.
chlorobenzene
1,1-dichlorobenzene
1,2-dichlorobenzene
1,4-dichlorobenzene
Friedel Craft Alkylation Concept 3
Video transcript
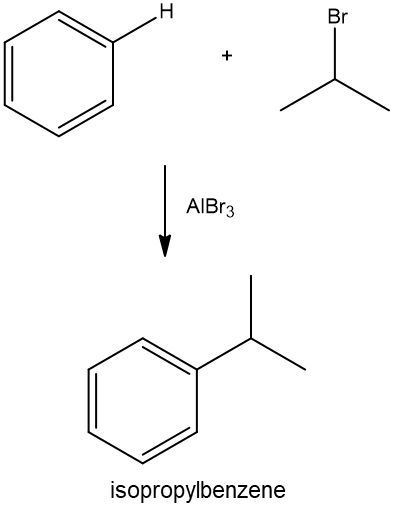
In this video, we're going to take a look at Friedel-Crafts Alkylation. Here in this reaction, benzene reacts with an alkyl halide, and what happens is that one of the hydrogens on the benzene ring is substituted with an alkyl group. To do this, a catalyst of Aluminum X3 must be used and has to contain the same halogen as the reagent. So basically, what we mean by this, if we take a look at this Friedel-Crafts reaction down below, is CH3X. This X could represent either bromine or chlorine. If it's bromine, then this has to be AlBr3. If this X is chlorine, then this has to be AlCl3. So, the halogens have to match between these two. Now, in this reaction, all that happens is that one of the hydrogens on benzene gets substituted out, and the alkyl portion of our alkyl halide comes in. The alkyl portion of our alkyl halide here would be methyl, so we end up making methylbenzene as our final answer.
Friedel Craft Alkylation Example 2
Video transcript
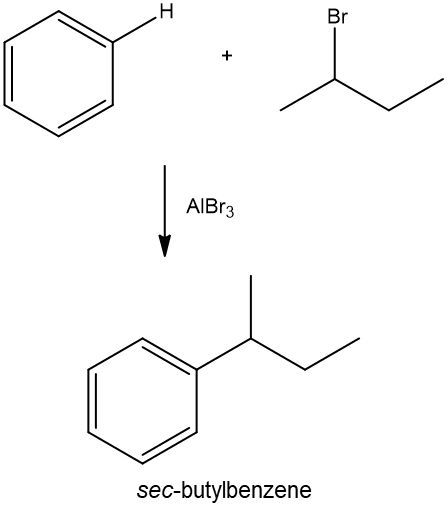
Name the major product of the following Friedel Crafts alkylation reaction. Alright. So here we have our Benzene, and we know we're still going to have Benzene at the end, so we can redraw it. And we know that essentially what happens is, one of the hydrogens on Benzene gets substituted out with the alkyl group portion of my alkyl halide. So here we have Ethyl Chloride and we have Aluminum Chloride as the catalyst. So here, we're going to attach Ethyl to my Benzene. This would be my major product form, and its name would be Ethylbenzene. So just remember with Friedel Crafts alkylation, we're just substituting out a hydrogen and bringing in an alkyl group.
Summary of Benzene Reactions Concept 4
Video transcript
Here we have a summary of our benzene reactions. We have halogenation, in it we have the reagent of X2, which could represent Br2 or Cl2. We use our catalyst to get things going. In essence what happens is that we substitute out one of the hydrogens on benzene with one of these halogens here. If this is Br2, then this has to be FeBr3. If this is Cl2, then this has to be FeCl3. At the end, the H is substituted out, the halogen comes in.
Next, we have Friedel-Crafts alkylation. Here we use as our reagent an alkyl halide. X again could be Cl or Br, but now we're using the catalyst of aluminum, and it could be either aluminum chloride or aluminum bromide. Here, we substitute out our hydrogen and bring in our alkyl group. Here if the X is Cl, then this is aluminum chloride. If the X here is bromine, then this is aluminum bromide. So remember, both of these reactions are quite similar, they use similar types of catalysts, and they're just substituting out an H. This one substitutes in a halogen. This one here an alkyl group.
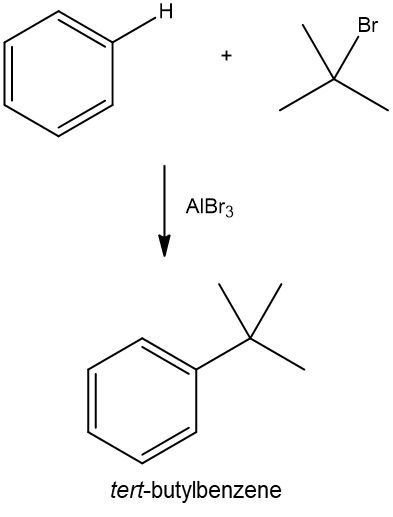
Provide a complete reaction of benzene Friedel Crafts alkylation with 2-bromo-2-methylpropane and name the product.
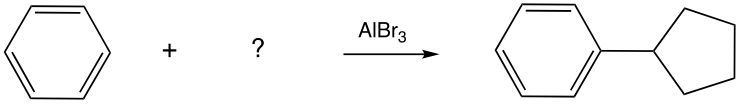
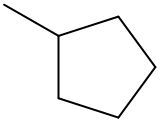
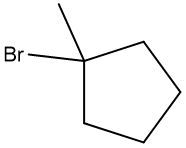
Fill in the missing reagent for the provided reaction.
Do you want more practice?
Your GOB Chemistry tutor
- Reaction of Br2 and FeBr3 with phenol can lead to three possible substitution products. Show the structure of ...
- Aromatic compounds do not normally react with hydrogen in the presence of a palladium catalyst but will if ver...
- The explosive trinitrotoluene (TNT) is made by carrying out three successive nitration reactions on toluene. I...
- Phenols undergo the same kind of substitution reactions that other aromatic compounds do (Section 13.11). Form...
- Ocimene, a compound isolated from the herb basil, has three double bonds and the IUPAC name 3,7-dimethyl-1, 3-...
- Cinnamaldehyde, the pleasant-smelling substance found in cinnamon oil, has the following structure:<IMAGE&g...