The structure of the amino acid lysine (in its uncharged form) is shown below.
b. Is lysine likely to be water-soluble? Explain.

 Verified step by step guidance
Verified step by step guidance


The structure of the amino acid lysine (in its uncharged form) is shown below.
b. Is lysine likely to be water-soluble? Explain.
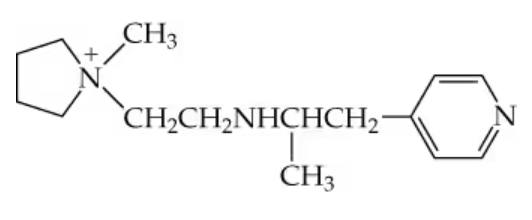
Write the structures of the following compounds:
a. Butyldiethylammonium bromide
Explain what bonds must be made or broken and where the electrons go when the hydrogen-bonded water between the two amines shown on page 507 reacts to form an amine, ammonium ion, and OH⁻.
Write the structure of benzylamine hydrochloride in two different ways, and name the hydrochloride as an ammonium salt.
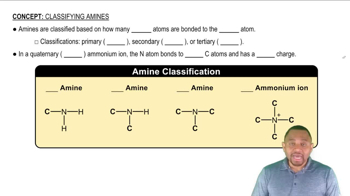
Which is the stronger base in each pair?
a. Ammonia or ethylamine
b.Triethylamine or pyridine
The structure of the amino acid lysine (in its uncharged form) is shown below.
a. Which amine groups would be able to participate in hydrogen bonding?