Are one or more of the disaccharides maltose, lactose, cellobiose, and sucrose part of the trisaccharide in Problem 20.23? If so, identify which disaccharide and its location. (Hint: Look for an α-1,4 link, β-1,4 link, or 1,2 link, and then determine if the correct monosaccharides are present.)

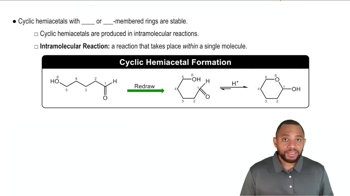
In solution, glucose exists predominantly in the cyclic hemiacetal form, which does not contain an aldehyde group. How is it possible for mild oxidizing agents to oxidize glucose?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Cyclic Hemiacetal Form
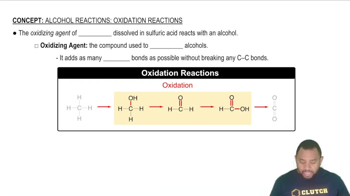
Oxidation of Alcohols
Reducing and Oxidizing Agents

Hydrolysis of both glycosidic bonds in the following trisaccharide A, B, C yields three monosaccharides.
c. Draw the Fischer projections for the three monosaccharides.
How many chiral carbon atoms are present in each of the molecules shown in Problem 20.31?
a.
b.
c.
d.
Classify the four carbohydrates (a)–(d) by indicating the nature of the carbonyl group and the number of carbon atoms present. For example, glucose is an aldohexose.
c.
Consider the trisaccharide A, B, C shown in Problem 20.23.
c. State the numbers of the carbon atoms that form glycosidic linkages between monosaccharide A and monosaccharide B.
Classify the four carbohydrates (a)–(d) by indicating the nature of the carbonyl group and the number of carbon atoms present. For example, glucose is an aldohexose.
d.
