Balance each of the following by adding coefficients, and identify the type of reaction for each:
a.

 Verified step by step guidance
Verified step by step guidance



Balance each of the following by adding coefficients, and identify the type of reaction for each:
a.
If purple spheres represent iodine atoms, white spheres represent hydrogen atoms, and all the molecules are gases,
a. write the formula for each of the reactants and products.
Balance each of the following by adding coefficients, and identify the type of reaction for each:
b.
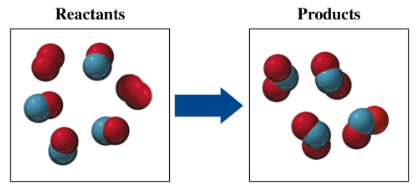
If red spheres represent oxygen atoms, blue spheres represent nitrogen atoms, and all the molecules are gases,
b. write a balanced equation for the reaction.
Ibuprofen, an anti-inflammatory drug in Advil, has the formula C13H18O2.
<IMAGE>
d. How many moles of ibuprofen contain 1.22 × 1023 atoms of C?
If red spheres represent oxygen atoms, blue spheres represent nitrogen atoms, and all the molecules are gases,
c. indicate the type of reaction as combination, decomposition, single replacement, double replacement, or combustion.