10. Acids and Bases
Strong Acid Strong Base Titrations (Simplified)
Problem 53
Textbook Question
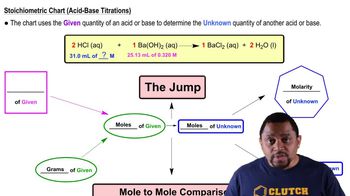
Textbook QuestionIf 32.8 mL of a 0.162 M NaOH solution is required to titrate 25.0 mL of a solution of H₂SO₄, what is the molarity of the H₂SO₄ solution? H₂SO₄(aq) + 2KOH(aq) → 2H₂O(l) + K₂SO₄(aq)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
169
views
Was this helpful?
Related Videos
Related Practice