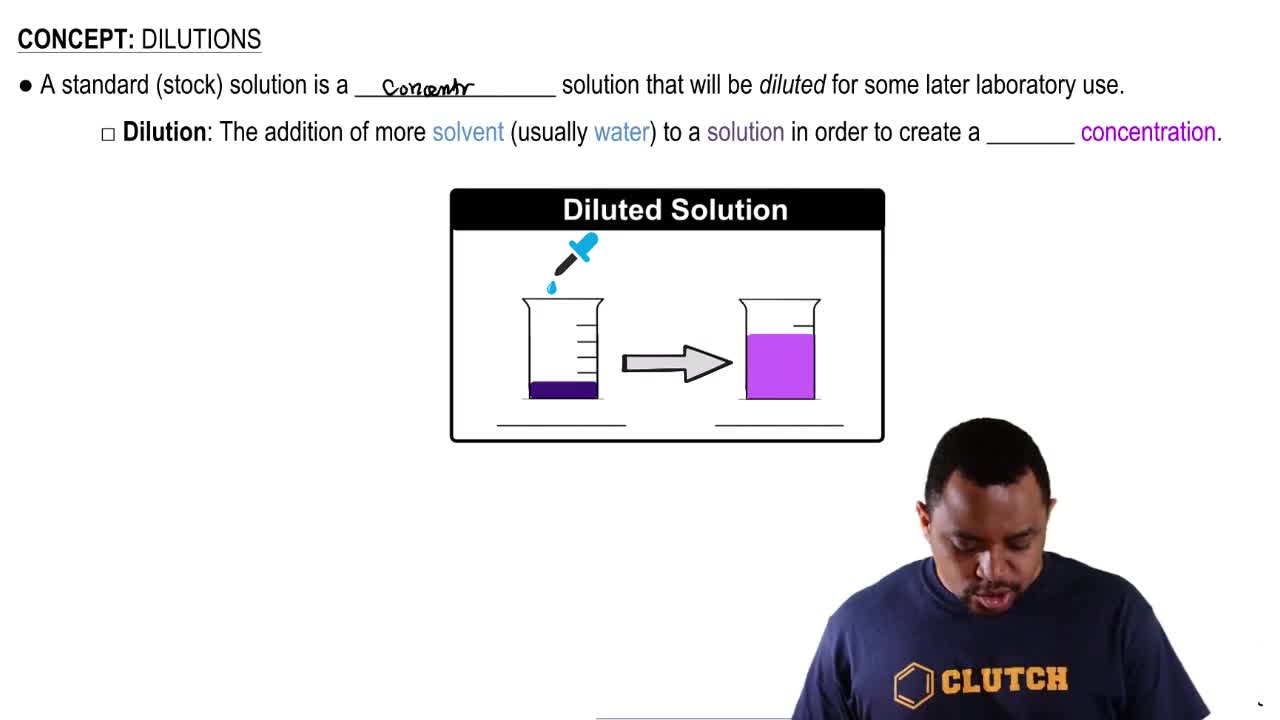
9. Solutions
Dilutions
Problem 113
Textbook Question
Textbook QuestionIn a laboratory experiment, a 10.0-mL sample of NaCl solution is poured into an evaporating dish with a mass of 24.10 g. The combined mass of the evaporating dish and NaCl solution is 36.15 g. After heating, the evaporating dish and dry NaCl have a combined mass of 25.50 g. (9.4) c. If water is added to 10.0 mL of the initial NaCl solution to give a final volume of 60.0 mL, what is the molarity of the diluted NaCl solution?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
161
views
Was this helpful?
Related Videos
Related Practice