10. Acids and Bases
Strong Acid Strong Base Titrations (Simplified)
Problem 112
Textbook Question
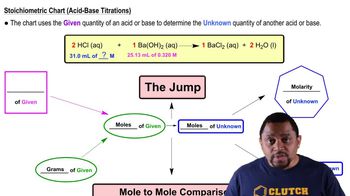
Textbook QuestionThe daily output of stomach acid (gastric juice) is 1000 mL to 2000 mL. Prior to a meal, stomach acid (HCl) typically has a pH of 1.42. (10.6, 10.7) c. The antacid milk of magnesia contains 400. mg of Mg(OH)₂ per teaspoon. Write the neutralization equation, and calculate the number of milliliters of stomach acid neutralized by 1 tablespoon of milk of magnesia (1tablespoon = 3teaspoons).
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
9mPlay a video:
253
views
Was this helpful?
Related Videos
Related Practice