7. Energy, Rate and Equilibrium
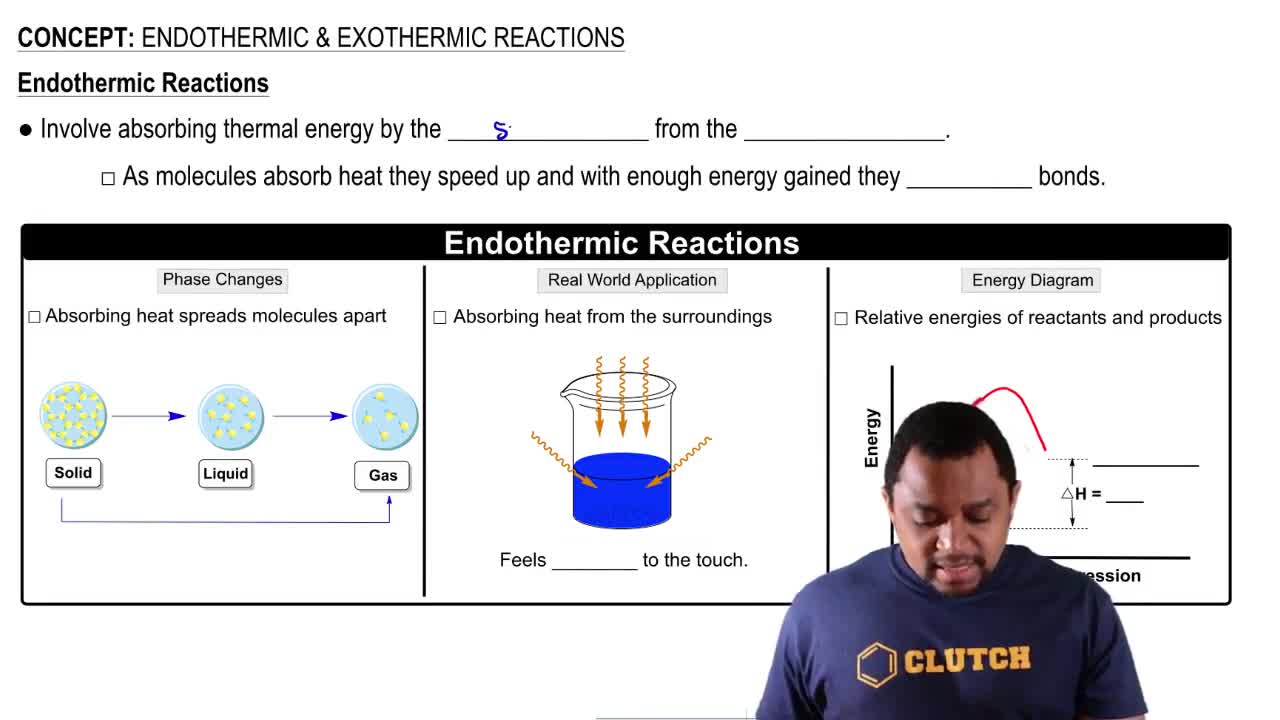
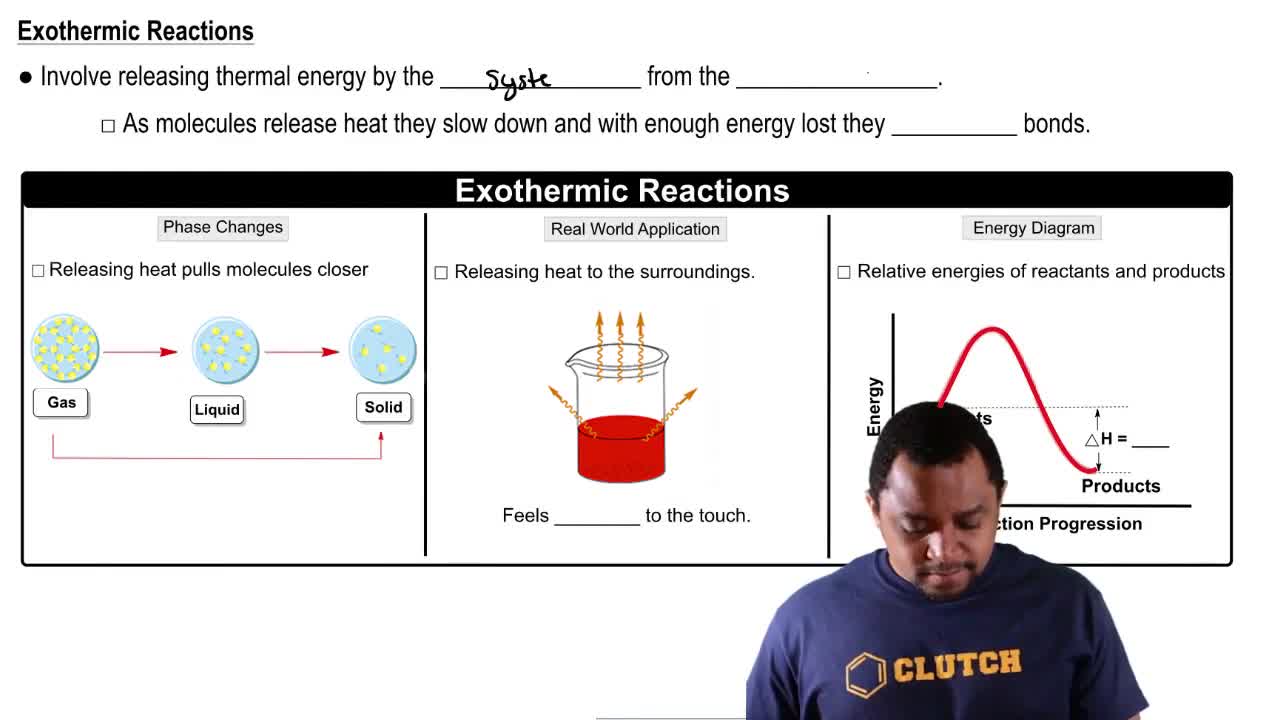
Endothermic & Exothermic Reactions
Problem 60
Textbook Question
Textbook QuestionHydrogen chloride can be made from the reaction of chlorine and hydrogen: Cl2(g) + H2(g) → 2 HCl(g) For this reaction, K = 26 x 10^33 and ∆H = -44 kcal/mol(-184 kJ/mol) at 25 °C. Is the reaction endothermic or exothermic?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
722
views
Was this helpful?
Related Videos
Related Practice