Identify the functional groups in cocaine

Complete the following equations (hint: remember that a nitrogen with three groups bound to it has a lone pair and one with four does not):
a. 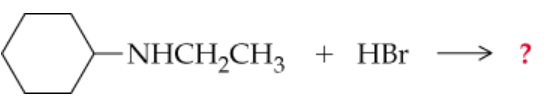
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Valence Shell Electron Pair Repulsion (VSEPR) Theory

Hybridization
Lone Pairs
Many hair conditioners contain an ammonium salt such as the following to help prevent 'fly-away' hair. These ions will react with neither acid nor base. Provide a reason why.
Draw the structures of the ammonium ions formed when the amines in Problem 16.30 are treated with acid.
a. N-Methylpentylamine
b. N-Ethylcyclobutylamine
c. p-Propylaniline
Choline has the following structure. Do you think that this substance reacts with aqueous hydrochloric acid? If so, what is the product? If not, why not?
Give names or structures for the following ammonium salts. Indicate whether each is the ammonium salt of a primary, secondary, or tertiary amine.
c. N-Butyl-N-isopropylhexylammonium chloride
Complete the following equations. (Hint: Remember that a nitrogen with three groups bound to it has a lone pair and one with four does not)
a.
