Textbook Question
Expand the following condensed structures into the correct structural formulas.
c. CH3CH2OCH2Cl
2035
views

 Verified step by step guidance
Verified step by step guidance


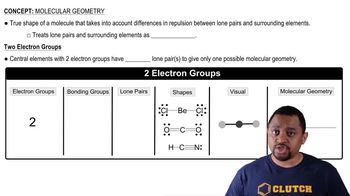
Expand the following condensed structures into the correct structural formulas.
c. CH3CH2OCH2Cl
Draw a Lewis structure for the following polyatomic ions:
b. Sulfite, SO32–
Distinguish between the following:
c. A lone pair and a shared pair of electrons
Consider the following possible structural formulas for C3H6O2. If a structure is not reasonable, explain what changes could be made to convert it to a reasonable structure.
a.
Based on electronegativity differences, would you expect bonds between the following pairs of atoms to be largely ionic or largely covalent?
b. Ca and Cl
Sketch the three-dimensional shape of the following molecules:
a. Methylamine, CH3NH2