6. Chemical Reactions & Quantities
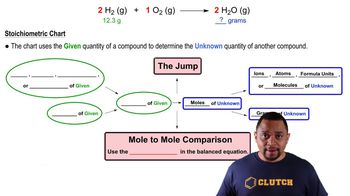
Stoichiometry
Problem 43b
Textbook Question
Textbook QuestionTitanium metal is obtained from the mineral rutile, TiO2. The process requires multiple steps, as shown in the following reactions: TiO2(S) + 2 Cl2(G) + 2 C(s) → TiCl4(s) + 2 CO(g) TiCl4(s) + 2 Mg(s) → Ti(s) + 2 MgCl2(s) How many kilograms of rutile are needed to produce 95 kg of Ti?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
234
views
Was this helpful?
Related Videos
Related Practice