How does an irreversible inhibitor function differently than a reversible inhibitor?

Problems 10.94 and 10.95 both mention enzymes that hydrolyze peptide bonds. How do you account for the fact that pepsin has a high catalytic activity at pH 1.5 but chymotrypsin has very little activity at pH 1.5?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Enzyme Catalysis
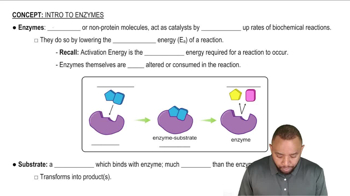
pH and Enzyme Activity

Specificity of Enzymes

Insulin is a protein hormone that functions as two polypeptide chains whose amino acid sequences are as follows:
A chain: GIVEQCCTSICSLTQLENYCN
B chain: FVNQHLCGDHLVEALYLV CGERGFFYTPKT
b. Considering the amino acid sequences, suggest how these two polypeptide chains might be held together in an active insulin molecule.
If each of the following amino acid side chains is present in the active site of an enzyme, indicate whether it would (a) serve a catalytic function, (b) serve to hold the substrate, or (c) both.
d. lysine
Pepsin, an enzyme that hydrolyzes peptide bonds in proteins, functions in the stomach at a pH optimum of 1.5 to 2.0. How is the rate of a pepsin-catalyzed reaction affected by each of the following conditions?
c. running the reaction at 0 °C
Pepsin, an enzyme that hydrolyzes peptide bonds in proteins, functions in the stomach at a pH optimum of 1.5 to 2.0. How is the rate of a pepsin-catalyzed reaction affected by each of the following conditions?
a. increasing the concentration of proteins
Meats spoil due to the action of enzymes that degrade the proteins. Fresh meats can be preserved for long periods of time by freezing them. Explain how freezing meats works to prevent spoilage.
