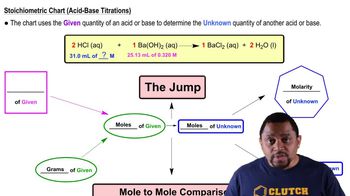
10. Acids and Bases
Strong Acid Strong Base Titrations (Simplified)
Problem 111
Textbook Question
Textbook QuestionOne of the most acidic lakes in the United States is Little Echo Pond in the Adirondacks in New York. Recently, this lake had a pH of 4.2, well below the recommended pH of 6.5. (10.6, 10.7) c. One way to raise the pH (and restore aquatic life) is to add limestone (CaCO₃). How many grams of CaCO₃ are needed to neutralize 1.0 kL of the acidic water from Little Echo Pond if the acid is sulfuric acid? H₂SO₄(aq) + CaCO₃(s) → CO₂(g) + H₂O(l) + CaCO₄(aq)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
229
views
Was this helpful?
Related Videos
Related Practice