Textbook Question
Complete the following equations:
a.
1281
views

 Verified step by step guidance
Verified step by step guidance
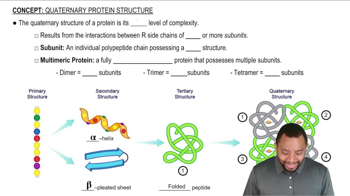


Complete the following equations:
a.
Write the structure of benzylamine hydrochloride in two different ways, and name the hydrochloride as an ammonium salt.
Which is the stronger base in each pair?
a. Ammonia or ethylamine
b.Triethylamine or pyridine
Complete the following equations:
c.
The structure of the amino acid lysine (in its uncharged form) is shown below.
a. Which amine groups would be able to participate in hydrogen bonding?
a. For the compound above, identify each nitrogen as either a primary, secondary, tertiary, quaternary, or aromatic amine.