Textbook Question
Identify the type of particle or radiation for each of the following:
b. 11H
2350
views

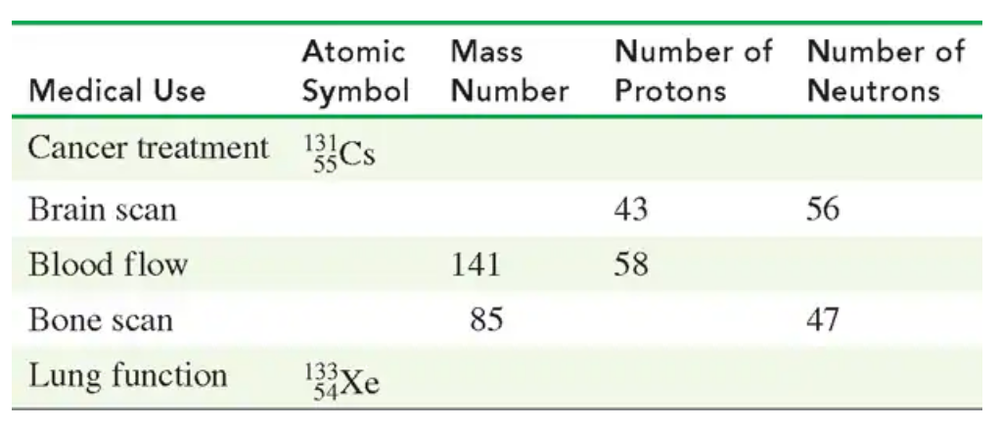
 Verified step by step guidance
Verified step by step guidance


Identify the type of particle or radiation for each of the following:
b. 11H
Identify each of the following:
c. 10X
Match the type of radiation (1 to 3) with each of the following statements:
1. alpha particle
2. beta particle
3. gamma radiation
b. shielding protection includes lead or thick concrete
Match the type of radiation (1 to 3) with each of the following statements:
1. alpha particle
2. beta particle
3. gamma radiation
c. can be very harmful if ingested
Match the type of radiation (1 to 3) with each of the following statements:
1. alpha particle
2. beta particle
3. gamma radiation
c. travels only a short distance in air