3. Ionic Compounds
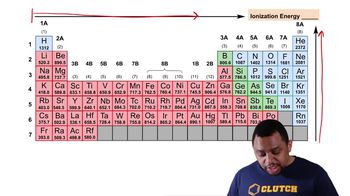
Periodic Trend: Ionization Energy (Simplified)
Problem 109
Textbook Question
Textbook QuestionWhy is the ionization energy of Ca higher than K, but lower than that of Mg? (4.7)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
262
views
Was this helpful?
Related Videos
Related Practice