8. Gases, Liquids and Solids
Dalton's Law: Partial Pressure (Simplified)
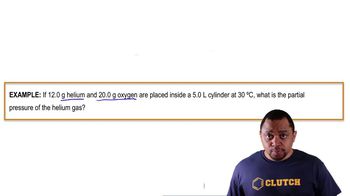
Problem 54
Textbook Question
Textbook QuestionSuppose a mixture contains helium and oxygen gases. If the partial pressure of helium is the same as the partial pressure of oxygen, what do you know about the number of helium atoms compared to the number of oxygen molecules? Explain.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
182
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 6 videos