7. Energy, Rate and Equilibrium
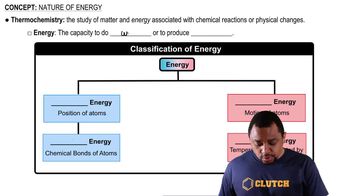
Nature of Energy
Problem 67
Textbook Question
Textbook QuestionFor the unbalanced combustion reaction shown, 1 mol of ethanol, C2H5OH, releases 327 kcal (1370 kJ): C2H5OH + O2 → CO2 + H2O If the density of ethanol is 0.789 g/mL, calculate the combustion energy of ethanol in kilocalories/milliliter and kilojoules/milliliter
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
7mPlay a video:
407
views
Was this helpful?
Related Videos
Related Practice