Find the pH of each mixture of acids. b. 0.150 M in HNO2 and 0.085 M in HNO3 c. 0.185 M in HCHO2 and 0.225 M in HC2H3O2 d. 0.050 M in acetic acid and 0.050 M in hydrocyanic acid
Ch.16 - Acids and Bases
Chapter 16, Problem 82a
For each strong base solution, determine [OH–], [H3O+], pH, and pOH. a. 8.77×10–3 M LiOH
 Verified step by step guidance
Verified step by step guidance1
<strong>Step 1:</strong> Identify the strong base and its dissociation. LiOH is a strong base, which means it dissociates completely in water. The dissociation can be represented as: \[ \text{LiOH} \rightarrow \text{Li}^+ + \text{OH}^- \]. Therefore, the concentration of \( \text{OH}^- \) ions is equal to the concentration of the LiOH solution, which is \( 8.77 \times 10^{-3} \text{ M} \).
<strong>Step 2:</strong> Calculate the \( [\text{OH}^-] \). Since LiOH dissociates completely, \( [\text{OH}^-] = 8.77 \times 10^{-3} \text{ M} \).
<strong>Step 3:</strong> Use the ion product of water to find \( [\text{H}_3\text{O}^+] \). The ion product of water at 25°C is \( K_w = 1.0 \times 10^{-14} \). Use the formula \( [\text{H}_3\text{O}^+] = \frac{K_w}{[\text{OH}^-]} \) to find \( [\text{H}_3\text{O}^+] \).
<strong>Step 4:</strong> Calculate the pOH. Use the formula \( \text{pOH} = -\log[\text{OH}^-] \) to find the pOH of the solution.
<strong>Step 5:</strong> Calculate the pH. Use the relationship \( \text{pH} + \text{pOH} = 14 \) to find the pH of the solution.

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Video duration:
1mWas this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Strong Bases
Strong bases are substances that completely dissociate in water to produce hydroxide ions (OH-). Common examples include alkali metal hydroxides like lithium hydroxide (LiOH). The concentration of the base directly correlates to the concentration of OH- ions in solution, which is crucial for calculating pH and pOH.
Recommended video:
Guided course

Strong Acid-Strong Base Titration
pH and pOH Calculations
pH is a measure of the acidity of a solution, defined as the negative logarithm of the hydrogen ion concentration ([H3O+]). Conversely, pOH measures the basicity, defined as the negative logarithm of the hydroxide ion concentration ([OH-]). The relationship between pH and pOH is given by the equation pH + pOH = 14 at 25°C, allowing for the conversion between these two measures.
Recommended video:
Guided course
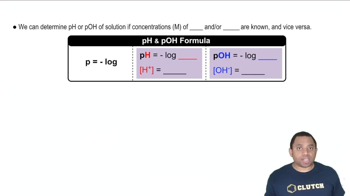
pH and pOH Calculations
Ion Product of Water
The ion product of water (Kw) is the equilibrium constant for the self-ionization of water, defined as Kw = [H3O+][OH-] = 1.0 x 10^-14 at 25°C. This relationship is essential for determining the concentrations of H3O+ and OH- in a solution, especially when dealing with strong bases, as it helps to find the corresponding [H3O+] when [OH-] is known.
Recommended video:
Guided course

Production of Hydrogen Example
Related Practice
Open Question
Open Question
Find the pH of each mixture of acids. a. 0.075 M in HNO3 and 0.175 M in HC7H5O2 b. 0.020 M in HBr and 0.015 M in HClO4 c. 0.095 M in HF and 0.225 M in HC6H5O d. 0.100 M in formic acid and 0.050 M in hypochlorous acid
Textbook Question
For each strong base solution, determine [OH–], [H3O+], pH, and pOH. a. 0.15 M NaOH b. 1.5×10–3 M Ca(OH)2 c. 4.8×10–4 M Sr(OH)2 d. 8.7×10–5 M KOH
681
views
Textbook Question
For each strong base solution, determine [OH–], [H3O+], pH, and pOH. b. 0.0112 M Ba(OH)2
443
views
Textbook Question
For each strong base solution, determine [OH–], [H3O+], pH, and pOH. c. 1.9×10–4 M KOH
295
views
Textbook Question
For each strong base solution, determine [OH–], [H3O+], pH, and pOH. d. 5.0×10–4 M Ca(OH)2
375
views
