Sodium azide is a shock-sensitive compound that releases N2 upon physical impact. The compound is used in automobile airbags. The azide ion is N3-. (a) Draw the Lewis structure of the azide ion that minimizes formal charge (it does not form a triangle). Is it linear or bent?
In ozone, O3, the two oxygen atoms on the ends of the molecule are equivalent to one another. (d) How many electrons are delocalized in the p system of ozone?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Delocalized Electrons
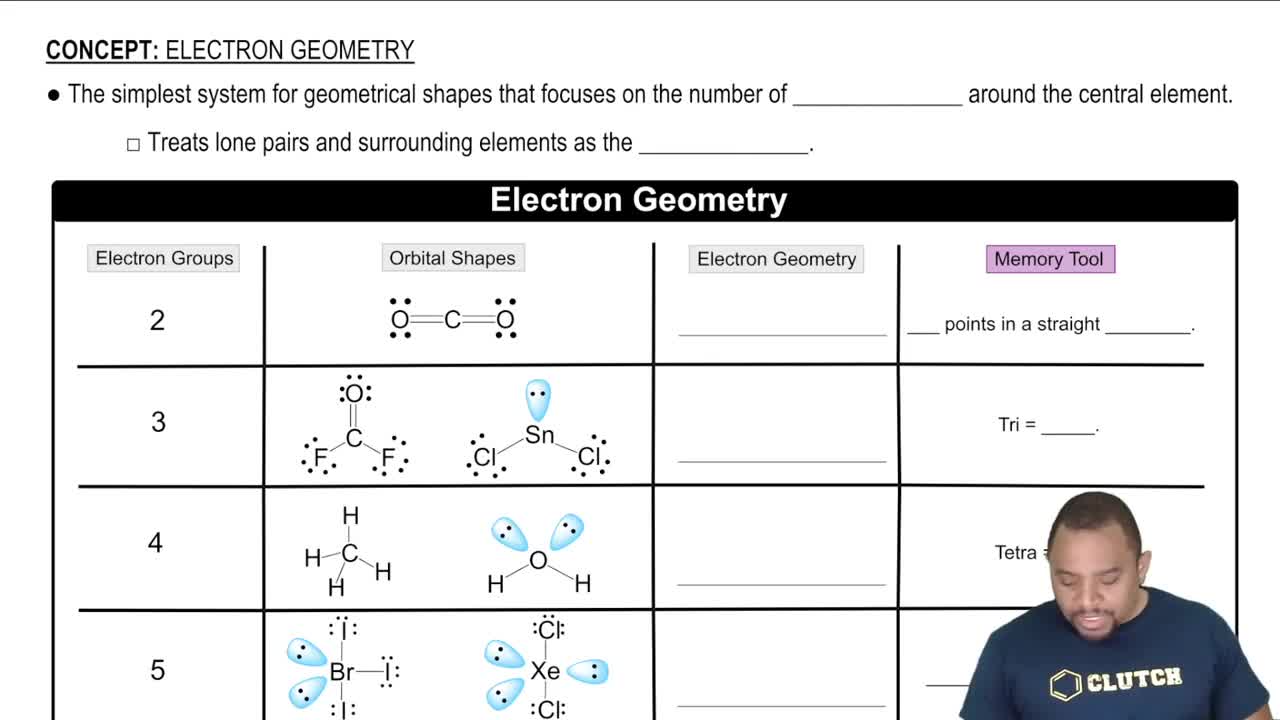
Resonance Structures

Molecular Orbital Theory

Sodium azide is a shock-sensitive compound that releases N2 upon physical impact. The compound is used in automobile airbags. The azide ion is N3-. (b) State the hybridization of the central N atom in the azide ion.
Butadiene, C4H6, is a planar molecule that has the following carbon–carbon bond lengths:
(a) Predict the bond angles around each of the carbon atoms and sketch the molecule.
Butadiene, C4H6, is a planar molecule that has the following carbon–carbon bond lengths:
(b) From left to right, what is the hybridization of each carbon atom in butadiene?
Butadiene, C4H6, is a planar molecule that has the following carbon–carbon bond lengths:
(c) The middle C¬C bond length in butadiene (1.48 Å) is a little shorter than the average C¬C single bond length (1.54 Å). Does this imply that the middle C¬C bond in butadiene is weaker or stronger than the average C¬C single bond?
