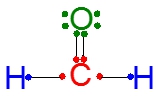
The octet rule is a fundamental concept in chemistry that describes the tendency of main group elements to achieve a stable electron configuration by having eight electrons in their outermost shell, similar to that of noble gases. This stability is often achieved through chemical bonding, where elements share or transfer electrons. In covalent bonding, two elements share a pair of valence electrons, which contributes to each element's total count of octet electrons.
Valence electrons are the outermost electrons of an atom and are determined by the element's group number in the periodic table. For instance, aluminum, which is in group 3A, has three valence electrons. When elements form bonds, they can gain shared electrons, which are the electrons contributed by another atom during the bonding process. The total number of octet electrons for an element is the sum of its valence electrons and the shared electrons it acquires through bonding.
To satisfy the octet rule, most main group elements aim for a total of eight octet electrons. However, hydrogen is an exception; it only requires two electrons to achieve the stable electron configuration of helium, thus it does not conform to the octet rule in the traditional sense. Understanding these concepts of valence, shared, and octet electrons is crucial for grasping the behavior of elements in chemical reactions and bonding scenarios.