15. Chemical Kinetics
Instantaneous Rate
15. Chemical Kinetics
Instantaneous Rate - Video Tutorials & Practice Problems

Get help from an AI Tutor
Ask a question to get started.
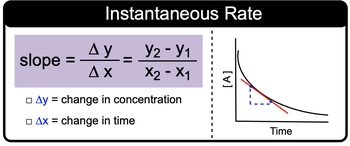
Instantaneous Rate is the rate a reaction at any particular point in time.
Instantaneous Rate
1
concept
Instantaneous Rate Concept 1
Video duration:
1mPlay a video:
2
example
Instantaneous Rate Example 1
Video duration:
1mPlay a video:
3
Problem
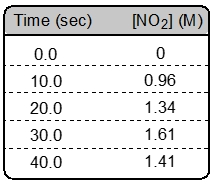
ProblemConsider the decomposition of dinitrogen pentoxide:2 N2O5 (g) → 4 NO2 (g) + O2 (g)
What is the instantaneous rate of this reaction at 20 seconds?
A
0.0325 M/s
B
0.0270 M/s
C
0.0380 M/s
D
0.015 M/s
Do you want more practice?
More setsYour General Chemistry tutor
Additional resources for Instantaneous Rate
PRACTICE PROBLEMS AND ACTIVITIES (11)
- (c) As a reaction proceeds, does the instantaneous reaction rate increase or decrease?
- The isomerization of methyl isonitrile 1CH3NC2 to acetonitrile 1CH3CN2 was studied in the gas phase at 215 C, ...
- The rate of disappearance of HCl was measured for the following reaction: CH3OH1aq2 + HCl1aq2¡CH3Cl1aq2 + H2O1...
- Consider the reaction: H2( g) + Br2( g) ¡ 2 HBr( g) The graph shows the concentration of Br2 as a function o...
- Consider the reaction: H2( g) + Br2( g) ¡ 2 HBr( g) The graph shows the concentration of Br2 as a function of ...
- Consider the reaction: 2 H2O2(aq) ¡ 2 H2O(l ) + O2( g) The graph shows the concentration of H2O2 as a function...
- Use the information in Table 14.1 and Figure 14.1 to estimate the instantaneous rate of appearance of NO2 at ...
- From the plot of concentration–time data in Figure 14.1, estimate: (b) the initial rate of decomposition of ...
- From the plot of concentration–time data in Figure 14.1, estimate: (a) the instantaneous rate of decomposition...
- From a plot of the concentration–time data in Worked Example 14.9, estimate: (b) the initial rate of decompo...
- From a plot of the concentration–time data in Worked Example 14.9, estimate: (a) the instantaneous rate of d...