12. Molecular Shapes & Valence Bond Theory
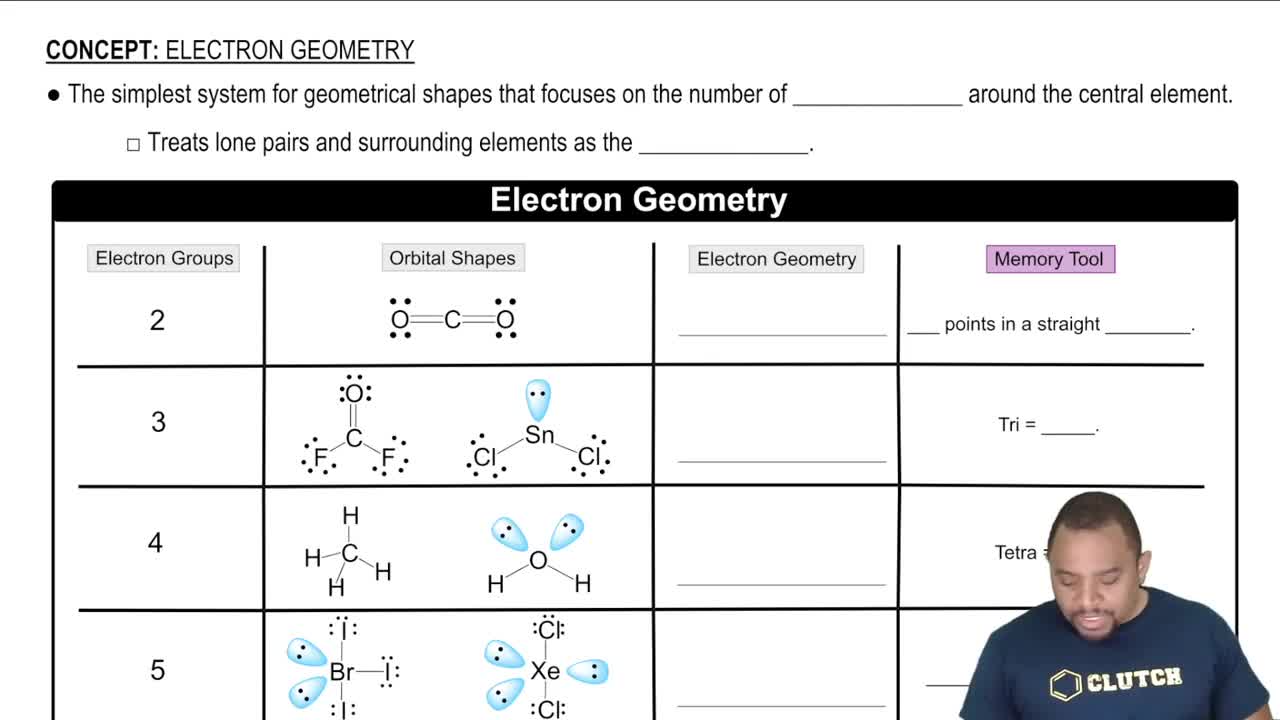
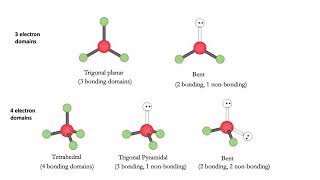
Electron Geometry
12. Molecular Shapes & Valence Bond Theory
Electron Geometry
Additional 1 creators.
Learn with other creators
Practice this topic
- Multiple Choice
Determine the electron geometry for the carbon disulfide molecule, CS2.
A. Linear
B. Bent
C. Trigonal planar
D. Tetrahedral
1078views3rank - Multiple Choice
Determine the number of electron groups for the following cation:AsBr2+.
A. 2
B. 3
C. 4
D. 1
1177views5rank1comments - Open Question
Draw and determine the electron geometry for the following molecule:TeBr4
699views8rank1comments - Multiple Choice
Determine the electron geometry of the nitrogen atom within methylamine, CH3NH2.
A. Linear
B. Trigonal Pyramidal
C. Tetrahedral
D. Trigonal Planar
1098views4rank - Open Question
At least two different numbers of electron groups can result in a linear molecule. What are they?
350views - Open Question
How does adding a lone pair affect the position of existing atoms and lone pairs?
294views - Open Question
Draw the Lewis structure of HCN and then determine its electron domain and molecular geometries.
790views - Multiple ChoiceAn AB4 molecule has one lone pair of electrons on the A atom (in addition to the four B atoms). What is the electron-domain geometry around the A atom?23views