- Download the worksheet to save time writing
- Start solving the practice problems
- If you're stuck, watch the video solutions
- See your summary to get more insights

A rock sample is suspected to be pure magnesium sulfate (MgSO4). It is pulverized and subjected to elemental analysis. The results revealed that the sample contains 21.45% Mg, 28.52% S, and 50.85% O. Is the sample of rock pure MgSO4?
Adam's catalyst, MO2, is used widely as a catalyst in organic synthesis reactions. Given that MO2 contains 14.1% oxygen by mass, determine the identity of M.
An oxide of chlorine with a partial molecular formula of Cl2Ox is analyzed. If the result showed that the compound contains 38.76% chlorine by mass, determine the value of x.
The empirical formula of a compound is PCl5 and it has a molar mass of 416.48 g/mol. Calculate the molecular formula.
Analysis of a compound shows that the compound contains twelve times as much carbon as hydrogen by mass. If the compound only contains carbon, hydrogen, bromine and oxygen, determine its molecular formula. Molar mass = 161 g/mol.
Consider the diagram below that illustrates an ionic compound. The cations are represented by light green spheres and the anions are represented by purple spheres. Choose among the following compounds that match the given illustration: Al2(C2O4)3, Ba(CH3COO)2, RbCl, Na2Cr2O7.

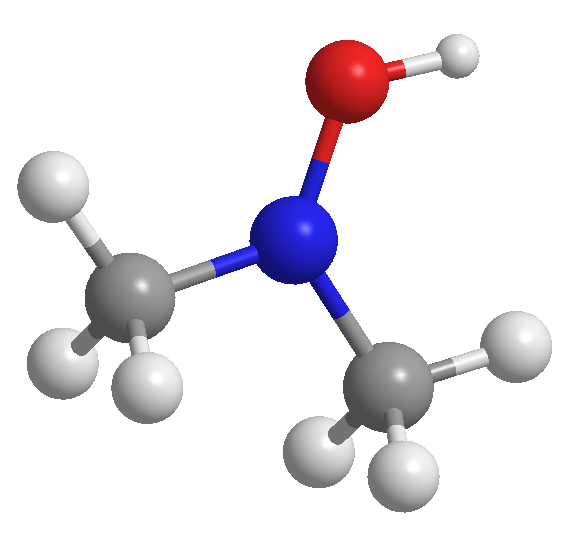
A 3D model of a compound is shown below. White spheres represent H, grey represents C, red represents O and blue represents N. Identify its molecular and structural formula.