- Download the worksheet to save time writing
- Start solving the practice problems
- If you're stuck, watch the video solutions
- See your summary to get more insights

The thermal dehydrogenation of propane is a first-order process and has a rate constant of 4.86×10−2 s−1 at 873 K:
C3H8(g) → C3H6(g) + H2(g)
Calculate the half-life of propane at 873 K.
The first-order reaction of A molecules (gray spheres) occurs in three vessels, 1, 2, and 3, of equal volume.
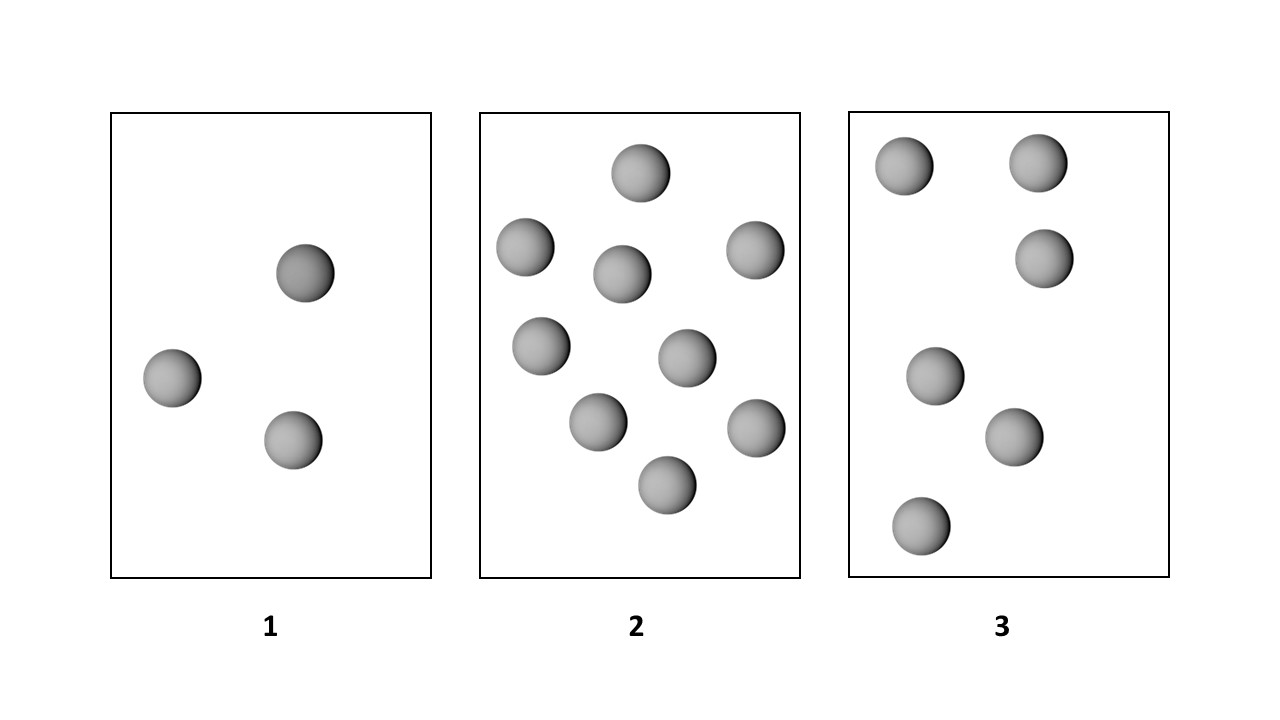 If the volume of each vessel is increased by a factor of 2, what will happen to the rates and half-lives of the reaction?
If the volume of each vessel is increased by a factor of 2, what will happen to the rates and half-lives of the reaction?
The first-order rate constant for the radioactive decay of Berkelium-248 (248Bk) and Seaborgium-260 (260Sg) is 0.0292 h−1 and 0.173 ms−1, respectively. Which sample decays at a faster rate?
The hypothetical gas-phase isomerization of AB → BA at 425°C was observed, and the data below was gathered.

What are its rate constant and half-life if the reaction is follows the first-order kinetics?
The components of a reaction after 0 mins and 20 mins are shown below.

After 7 half-life periods, what fraction of reactant is still present if the reaction is first-order?
The hydrolysis of the anticancer drug cisplatin occurs in first-order kinetics with a rate constant of 5.33 × 10−3 min−1 at 310.15 K. Assuming we start with 100% cisplatin, determine the time it will take for 35.0% of cisplatin to react? How about 50.0% of the cisplatin?
The half-life for the radioactive decay of U-238 is 4.5 billion years and is independent of initial concentration. How long will it take for 18% of the U-238 atoms in a sample of U-238 to decay?