- Download the worksheet to save time writing
- Start solving the practice problems
- If you're stuck, watch the video solutions
- See your summary to get more insights

What number of the following ions/atoms is/are paramagnetic?
Cr3+ Fe3+ Mg2+ Ne
Among the following subshells, 3d, 4s, 4p, 4d, 4f, 5s, 5p, 5d, 5f, and 6s, which can contain 4 electrons all with spin quantum number ms = –1/2?
Write a set of four quantum number that explain the electron in the last 2p orbital of the following orbital diagram. Arrange the quantum numbers as n, ℓ, mℓ, and ms.

Consider a hypothetical world in which the angular-momentum quantum number (l) can have integral values of l = 0, 1, 2, ..., n instead of l = 0, 1, 2, ..., n–1. The other three quantum numbers have the same possible values as they do in ours. Provide the orbital diagram for the element with an atomic number of 18.
For the element vanadium, V, Identify the electron configuration and the number of valence electrons in a neutral atom of vanadium.
Determine the number of inner, outer and valence electrons in an atom of the given elements?
a. O
b. C
c. Fe
d. Mg
e. S
Is gallium a metal, nonmetal, or metalloid? Why?
Rank the following elements from least metallic to most metallic.
Fr, Pb, Si, P, Ba, Se
Match each element with the correct sphere below.
a) Al atom
b) C atom
c) Cs atom

Given the atomic radius of the elements

Determine the atomic radius of Cs if the experimental bond length for Cs-Se is 4.18 Å
Arrange the following elements in descending order based on their atomic radii:
K, Pb, Br, Sn, and P
Which of the following options below show the isoelectronic ions in order of decreasing radii?
a. Cl – > Br – > F –
b. S2- > Cl – > Ar
c. Ca2+ > Na + > Ar
d. Ca2+ > Sr 2+ > Mg2+
LiCl, NaF, KBr, RbI
The experimentally measured cation-anion distances are: LiCl = 2.50 Å; NaF = 2.31 Å; KBr = 3.30 Å and RbI = 3.67 Å
Use the diagram below to determine the theoretical cation-anion distances for the ionic compounds above.
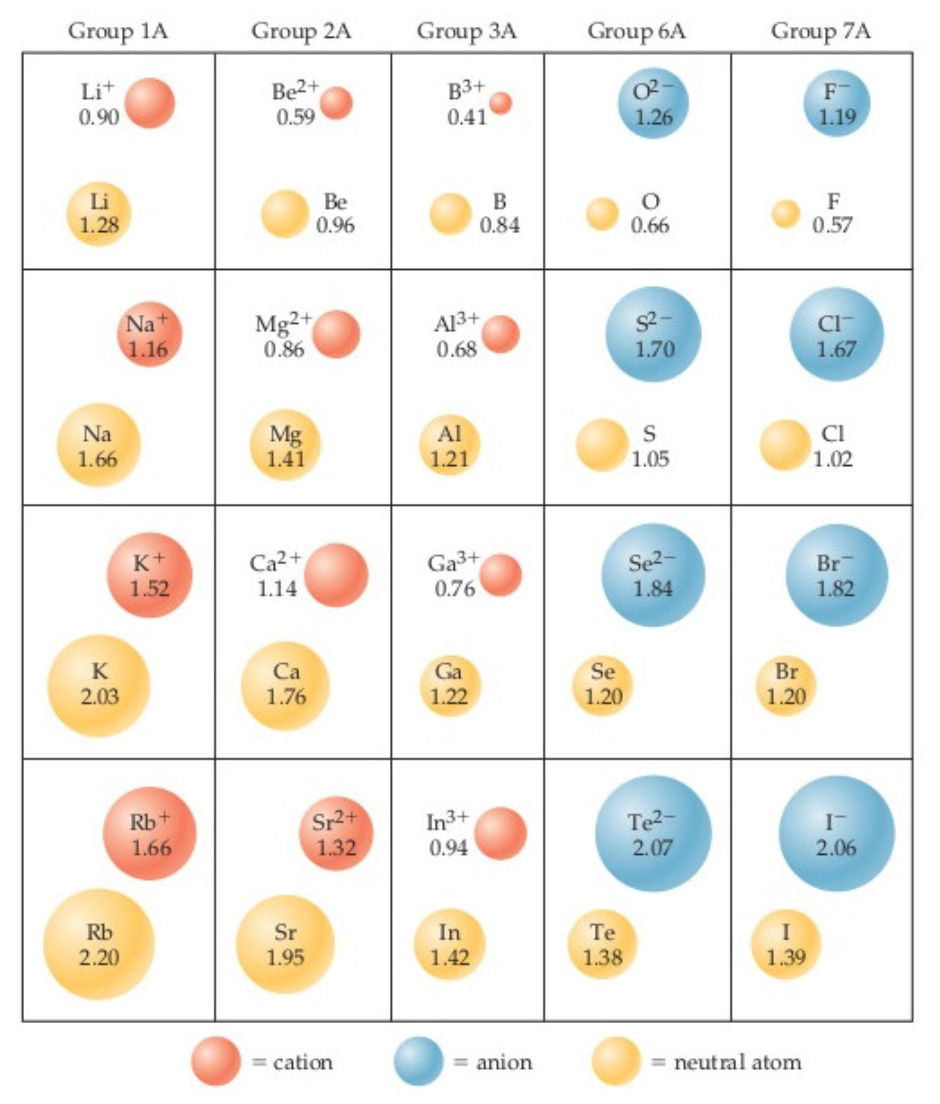
Which is the larger species in each pair?
a. Mg or Mg2+
b. Fe- or Fe2+
c. Nb- or Nb5+
d. Se or Se2-