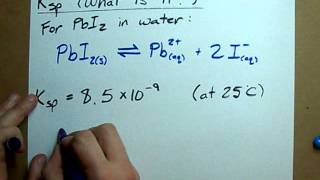18. Aqueous Equilibrium
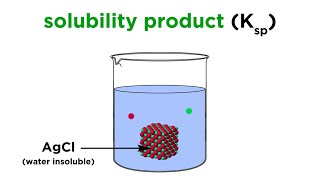
Solubility Product Constant: Ksp
Problem 89b
Textbook Question
Textbook QuestionUse the given molar solubilities in pure water to calculate Ksp for each compound. b. PbF2; molar solubility = 5.63 * 10- 3 M
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
2170
views
1
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos