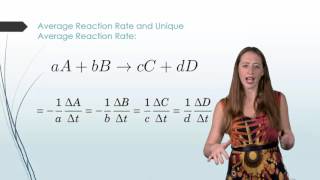15. Chemical Kinetics
Average Rate of Reaction
Problem 25d
Textbook Question
Textbook QuestionConsider the reaction: 2 HBr( g) ¡ H2( g) + Br2( g) b. In the first 25.0 s of this reaction, the concentration of HBr dropped from 0.600 M to 0.512 M. Calculate the average rate of the reaction during this time interval.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
2788
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos