8. Thermochemistry
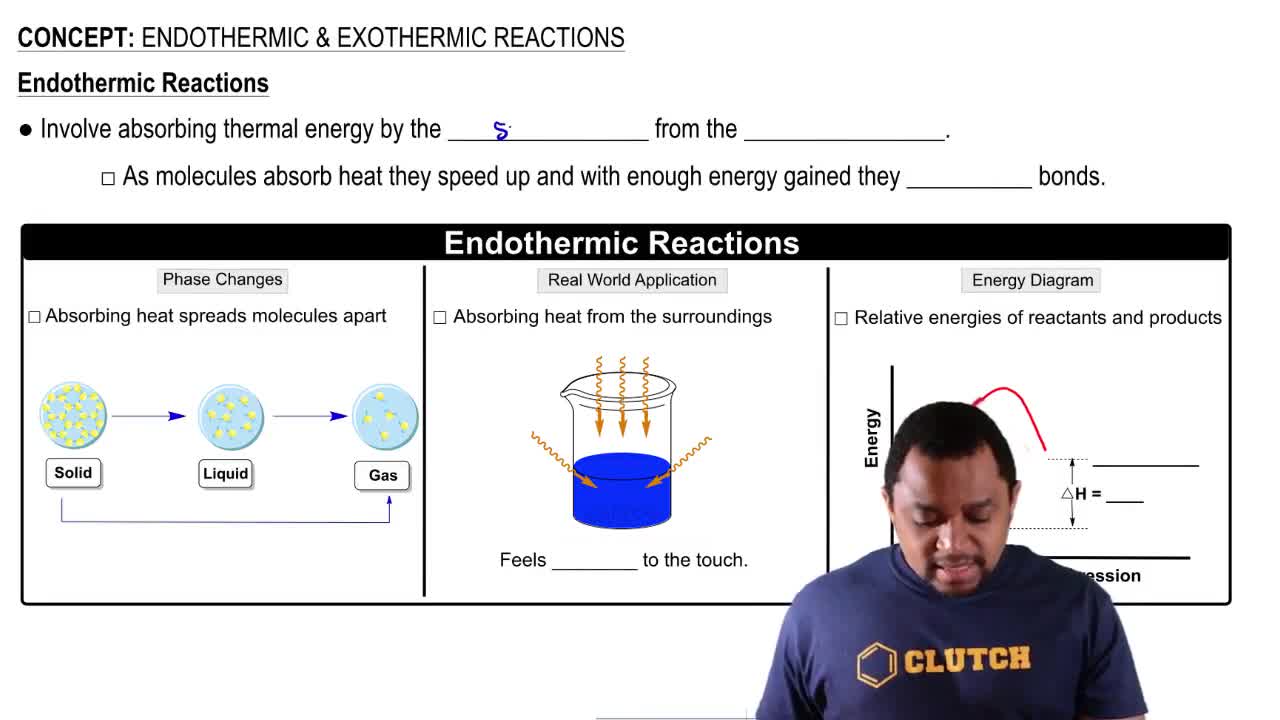
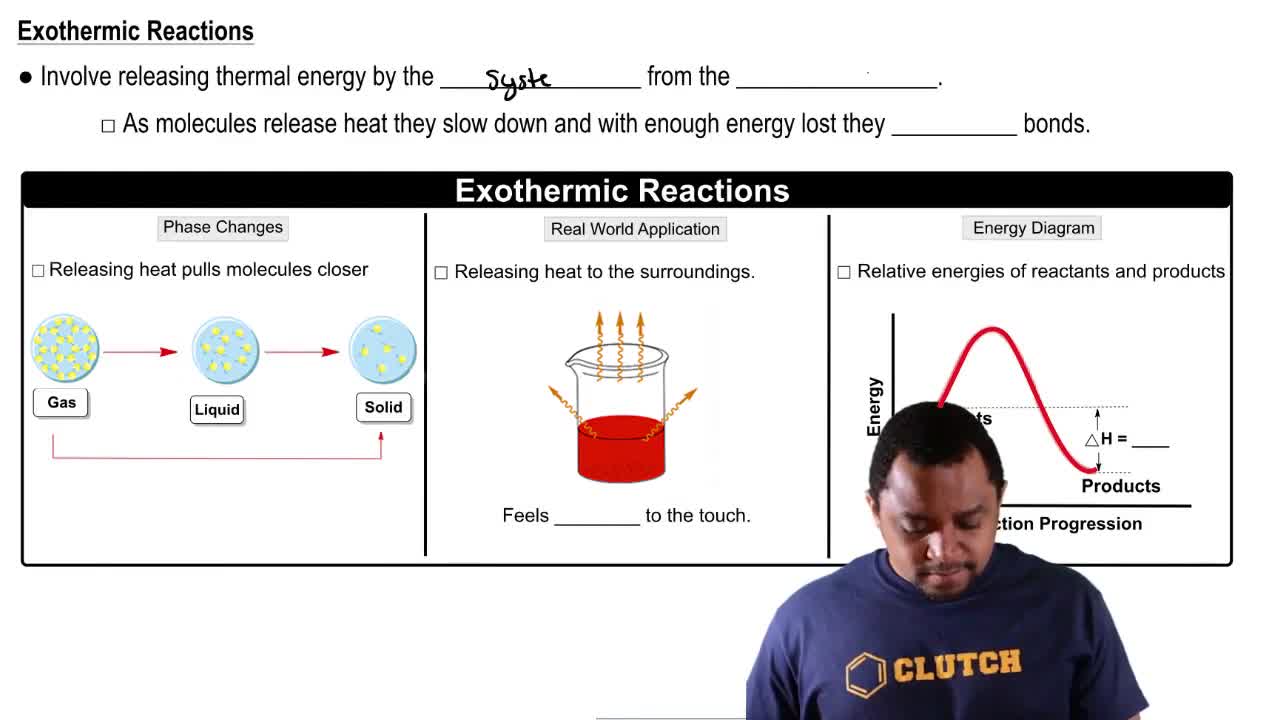
Endothermic & Exothermic Reactions
Problem 26a
Textbook Question
Textbook QuestionFor the following processes, calculate the change in internal energy of the system and determine whether the process is endothermic or exothermic: (a) A balloon is cooled by removing 0.655 kJ of heat. It shrinks on cooling, and the atmosphere does 382 J of work on the balloon.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
966
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos