20. Electrochemistry
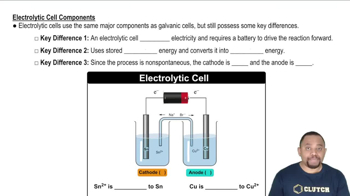
Electrolytic Cell
Problem 40b
Textbook Question
Textbook QuestionPorous pellets of TiO2 can be reduced to titanium metal at the cathode of an electrochemical cell containing molten CaCl2 as the electrolyte. When the TiO2 is reduced, the O2-ions dis-solve in the CaCl2 and are subsequently oxidized to O2 gas at the anode. This approach may be the basis for a less expensive process than the one currently used for producing titanium.

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
350
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 6 videos