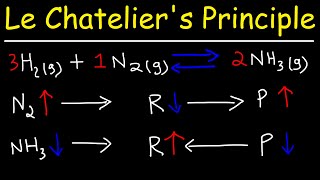
16. Chemical Equilibrium
Le Chatelier's Principle
Problem 72
Textbook Question
Textbook QuestionCoal can be used to generate hydrogen gas (a potential fuel) by the endothermic reaction: C(s) + H2O(g) ⇌ CO(g) + H2(g) If this reaction mixture is at equilibrium, predict whether each disturbance will result in the formation of additional hydrogen gas, the formation of less hydrogen gas, or have no effect on the quantity of hydrogen gas. e. adding a catalyst to the reaction mixture
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
48sPlay a video:
1282
views
1
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos