18. Aqueous Equilibrium
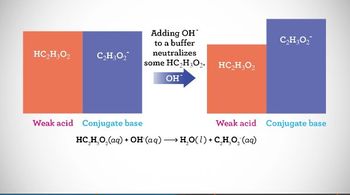
Intro to Buffers
Multiple Choice
Multiple ChoiceDetermine which of the following actions will destroy a buffer composed of 0.50 L of 1.44 M H3PO4 and 0.60 L of 1.25 M NaH2PO4−.
a) Addition of 1.45 moles of KH2PO4
b) Addition of 0.85 moles of HCl
c) Addition of water
d) Addition of 0.30 moles of Ca(OH)2
e) Addition of 0.70 moles of HIO4
A
Addition of 1.45 moles of KH2PO4
B
Addition of 0.85 moles of HCl
C
Addition of water
D
Addition of 0.30 moles of Ca(OH)2
E
Addition of 0.70 moles of HIO4
310
views
5
rank
Related Videos
Related Practice
Showing 1 of 16 videos