18. Aqueous Equilibrium
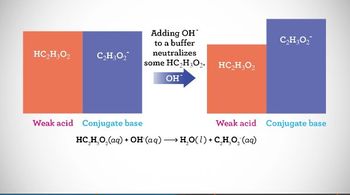
Intro to Buffers
Multiple Choice
Multiple ChoiceDetermine how many grams of sodium acetate, NaCH3CO2 (MW:82.05 g/mol), you would mix into enough 0.065 M acetic acid CH3CO2H (MW:60.05 g/mol) to prepare 3.2 L of a buffer with a pH of 4.58. The Ka is 1.8 x 10-5.
A
10.90 grams
B
11.68 grams
C
6.35 grams
D
2.21 grams
2683
views
9
comments
Related Videos
Related Practice
Showing 1 of 16 videos