19. Chemical Thermodynamics
Entropy
Problem 89
Textbook Question
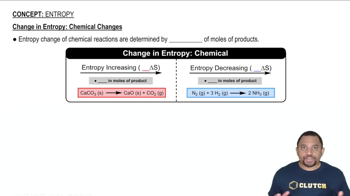
Textbook QuestionThe reaction 2 Mg1s2 + O21g2¡2 MgO1s2 is highly spontaneous. A classmate calculates the entropy change for this reaction and obtains a large negative value for ΔS °. Did your classmate make a mistake in the calculation? Explain.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
339
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos