17. Acid and Base Equilibrium
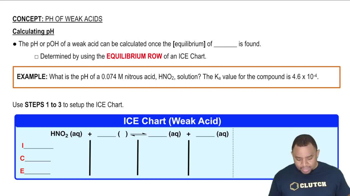
pH of Weak Acids
Problem 52
Textbook Question
Textbook QuestionPhenylacetic acid 1C6H5CH2COOH2 is one of the substances that accumulates in the blood of people with phenylketonuria, an inherited disorder that can cause mental retardation or even death. A 0.085 M solution of C6H5CH2COOH has a pH of 2.68. Calculate the Ka value for this acid.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
1284
views
1
comments
Was this helpful?
Related Videos
Related Practice
Showing 1 of 5 videos