8. Thermochemistry
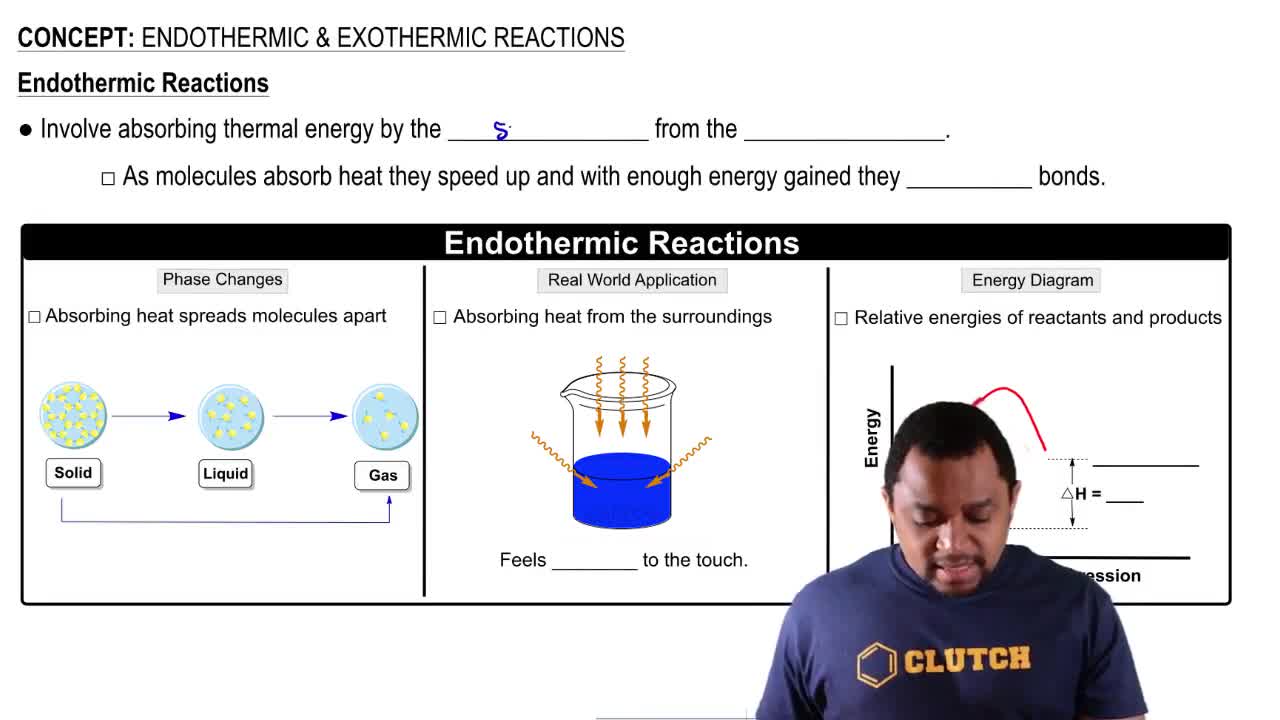
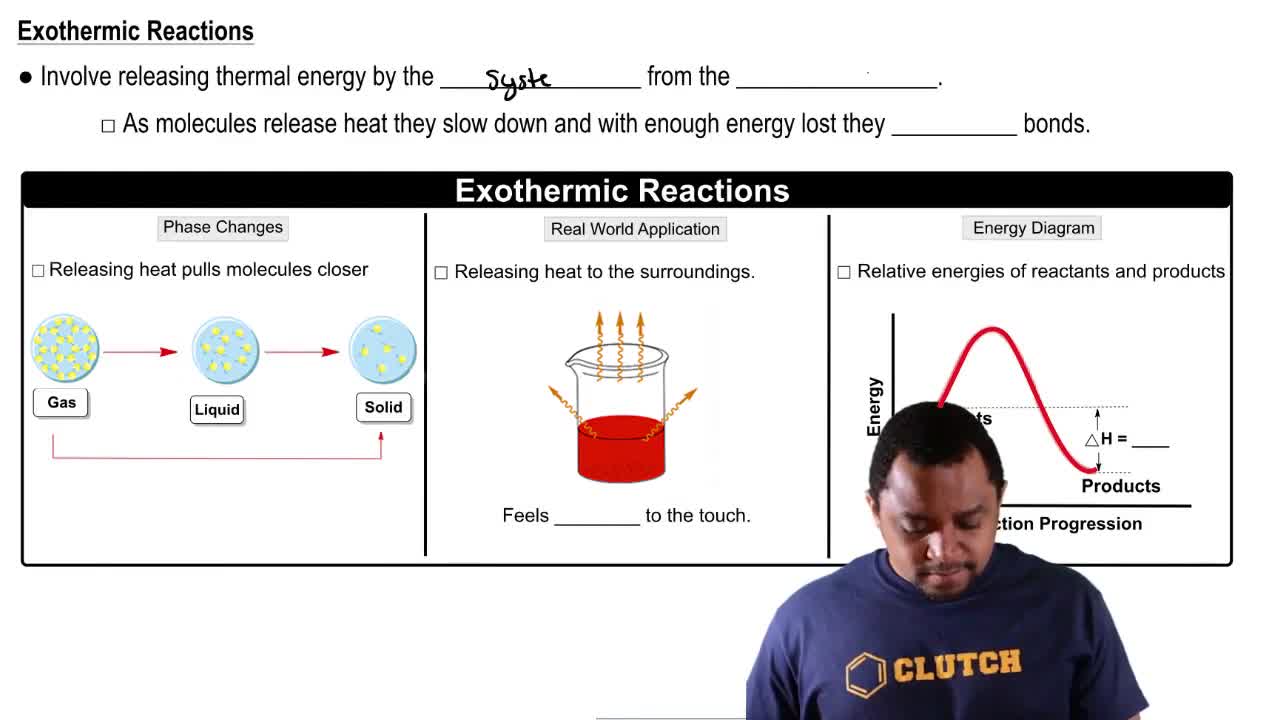
Endothermic & Exothermic Reactions
Problem 48a
Textbook Question
Textbook QuestionConsider the decomposition of liquid benzene, C6H61l2, to gaseous acetylene, C2H21g2: C6H61l2 ¡ 3 C2H21g2 H = +630 kJ (d) If C6H61g2 were consumed instead of C6H61l2, would you expect the magnitude of H to increase, decrease, or stay the same? Explain.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
572
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos