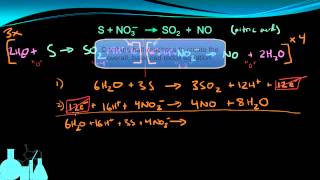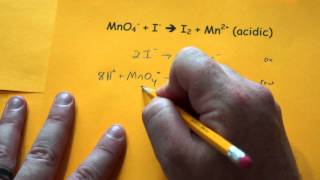6. Chemical Quantities & Aqueous Reactions
Balancing Redox Reactions: Acidic Solutions
Multiple Choice
Multiple ChoiceA 10.00-mL sample of a 0.130 M H3PO4 solution is added to 13.00 mL of a 0.150 M Ba(OH)2 solution. When the reaction is complete, what spectator ions are present?
2 H3PO4 (aq) + 3 Ba(OH)2 (aq) → Ba3(PO4)2 (s) + 6 H2O (l)
A
[H]+
B
[PO4]3–
C
[Ba]2+
D
[OH]–
E
All of the above ions are present.
F
No spectator ions are present.
212
views
Related Videos
Related Practice
Showing 1 of 8 videos