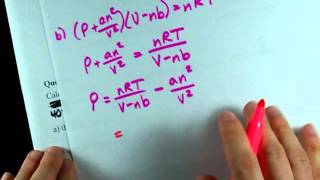7. Gases
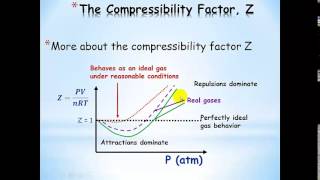
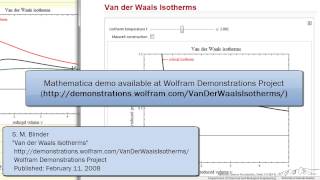
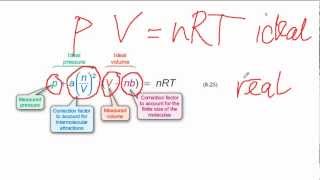
Van der Waals Equation
Problem 117
Textbook Question
Textbook QuestionLarge amounts of nitrogen gas are used in the manufacture of ammonia, principally for use in fertilizers. Suppose 120.00 kg of N21g2 is stored in a 1100.0-L metal cylinder at 280 °C. (b) By using the data in Table 10.3, calculate the pressure of the gas according to the van der Waals equation.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
7mPlay a video:
920
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos