10. Periodic Properties of the Elements
Periodic Trend: Ionization Energy
Open Question
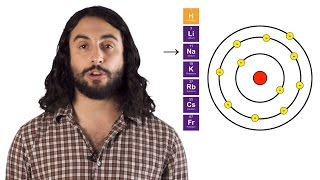
Open QuestionWrite a chemical equation representing the second ionization energy for lithium. Use e− as the symbol for an electron.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
216
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 14 videos