6. Chemical Quantities & Aqueous Reactions
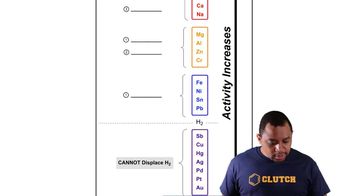
Activity Series
Problem 144
Textbook Question
Textbook QuestionAn unknown metal (M) was found not to react with either water or steam, but its reactivity with aqueous acid was not investigated. When a 1.000 g sample of the metal was burned in oxygen and the resulting metal oxide converted to a metal sulfide, 1.504 g of sulfide was obtained. What is the identity of the metal?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
403
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos