10. Periodic Properties of the Elements
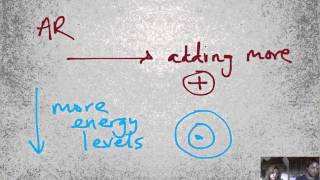
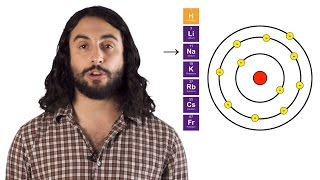
Periodic Trend: Ionic Radius
Problem 35
Textbook Question
Textbook QuestionProvide a brief explanation for each of the following.
K+ is larger than Na+.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
942
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos