2. Atoms & Elements
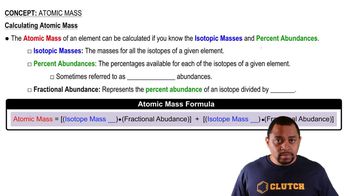
Atomic Mass
Multiple Choice
Multiple ChoiceNeon possesses three naturally occurring isotopes. 20Ne has a percent abundance of 90.48% and an isotopic mass of 19.99244 amu, 21Ne has a percent abundance of 0.27% and an isotopic mass of 20.99384 amu, and 22Ne has a percent abundance of 9.25%. What is the isotopic mass of the 22Ne isotope?
A
20.9182 amu
B
15.119 amu
C
23.001 amu
D
21.98768 amu
382
views
Related Videos
Related Practice
Showing 1 of 16 videos