17. Acid and Base Equilibrium
Triprotic Acids and Bases Calculations
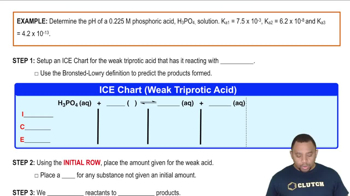
Problem 17.142
Textbook Question
Textbook QuestionThe acidity of lemon juice is derived primarily from citric acid (H3Cit), a triprotic acid. What are the concentrations of H3Cit, H2Cit-, HCit2-, and Cit3- in a sample of lemon juice that has a pH of 2.37 and a total concentration of the four citrate-containing species of 0.350 M? <IMAGE>
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
14mPlay a video:
102
views
Was this helpful?
Related Videos
Related Practice