18. Aqueous Equilibrium
Titrations: Strong Acid-Strong Base
Problem 81a
Textbook Question
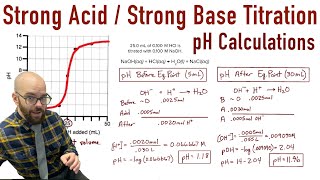
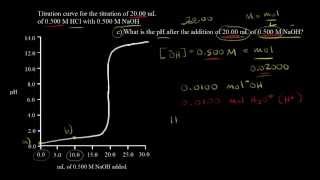
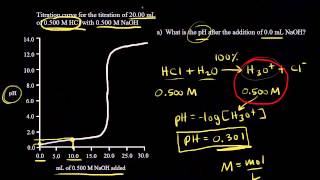
Textbook QuestionConsider the titration of 60.0 mL of 0.150 M HNO3 with 0.450 M NaOH. (a) How many millimoles of HNO3 are present at the start of the titration? (b) How many milliliters of NaOH are required to reach the equivalence point? (c) What is the pH at the equivalence point? (d) Sketch the general shape of the pH titration curve.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
825
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos