16. Chemical Equilibrium
ICE Charts
Problem 88
Textbook Question
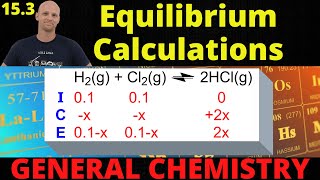
Textbook QuestionThe equilibrium constant for the reaction SO2(g) + NO2(g) ⇌ SO3(g) + NO(g) is Kc = 3.0. Find the amount of NO2 that must be added to 2.4 mol of SO2 in order to form 1.2 mol of SO3 at equilibrium.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
1912
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos