2. Atoms & Elements
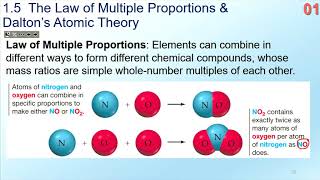
Law of Multiple Proportions
Problem 11b
Textbook Question
Textbook QuestionA 1.0-g sample of carbon dioxide (CO2) is fully decomposed into its elements, yielding 0.273 g of carbon and 0.727 g of oxygen. (b) If a sample of a different compound decomposes into 0.429 g of carbon and 0.571 g of oxygen, what is its ratio of the mass of O to C?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
813
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos