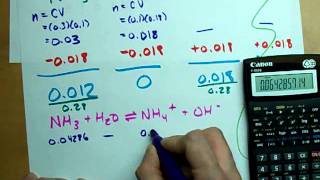18. Aqueous Equilibrium
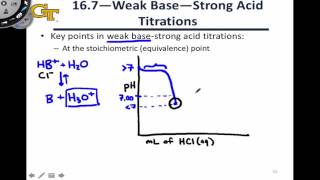
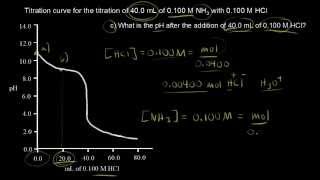
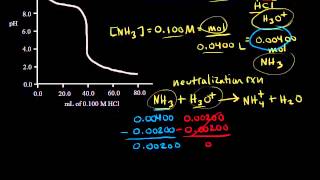
Titrations: Weak Base-Strong Acid
Multiple Choice
Multiple ChoiceA solution contains 100.0 mL of 0.550 M sodium nitrite, NaNO2. Find the pH after the addition of 180.0 mL of 0.400 M HClO4. The Ka of HNO2 is 4.6 × 10−4.
A
11.17
B
1.22
C
3.85
D
12.78
183
views
Related Videos
Related Practice
Showing 1 of 12 videos