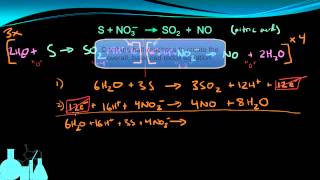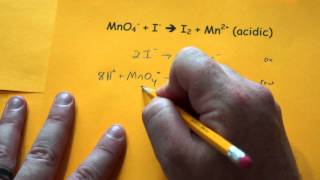6. Chemical Quantities & Aqueous Reactions
Balancing Redox Reactions: Acidic Solutions
Problem 158a
Textbook Question
Textbook QuestionOn heating a 0.200 g sample of a certain semimetal M in air, the corresponding oxide M2O3 was obtained. When the oxide was dissolved in aqueous acid and titrated with KMnO4, 10.7 mL of 0.100 M MnO4- was required for complete reac- tion. The unbalanced equation is X (b) How many moles of oxide were formed, and how many moles of semimetal were in the initial 0.200 g sample?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
337
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos